Dimensional analysis can also be used to solve certain types of partial differential equations. If this seems too good to be true, it isn't. Here we will concentrate on the solution of the diffusion equation; we will encounter this equation many times in the remainder of the course, so it will be useful to work out some of its properties now.
We'll start by deriving the one-dimensional diffusion, or heat, equation.![]() 1 Let
1 Let ![]() represent the temperature of a metal bar
at a point x at time t (I'll use
represent the temperature of a metal bar
at a point x at time t (I'll use ![]() to avoid confusion with the
symbol for the dimension of time, T). The first step is the derivation
of a continuity equation
for the heat flow
in the bar. Let the bar have a cross sectional area A, so that the
infinitesimal volume of the bar between x and
to avoid confusion with the
symbol for the dimension of time, T). The first step is the derivation
of a continuity equation
for the heat flow
in the bar. Let the bar have a cross sectional area A, so that the
infinitesimal volume of the bar between x and ![]() is
is ![]() . The quantity of heat contained in this volume is
. The quantity of heat contained in this volume is ![]() ,
with
,
with ![]() the specific heat at constant pressure per unit volume; it
has dimensions
the specific heat at constant pressure per unit volume; it
has dimensions ![]() . In a time interval dt
this heat changes by an amount
. In a time interval dt
this heat changes by an amount ![]() due to the change in temperature. This change in the heat must come from
somewhere, and is the result of a flux of heat q(x,t) through the area A
(q is the heat flowing through a unit area per unit time). Into the left
side of the volume an amount of heat q A dt flows in a time dt; on the
right hand side of the volume a quantity
due to the change in temperature. This change in the heat must come from
somewhere, and is the result of a flux of heat q(x,t) through the area A
(q is the heat flowing through a unit area per unit time). Into the left
side of the volume an amount of heat q A dt flows in a time dt; on the
right hand side of the volume a quantity ![]() flows out in a time dt, so that the net accumulation of heat
in the volume is
flows out in a time dt, so that the net accumulation of heat
in the volume is ![]() . Equating the two
expressions for the rate of change of the heat in the volume
. Equating the two
expressions for the rate of change of the heat in the volume ![]() ,
we find
,
we find
which is the equation of continuity. It is a mathematical expression of the
conservation of heat in the infinitesimal volume ![]() . We
supplement this with a phenomenological law of heat conduction, known as
Fourier's law:
the heat flux is proportional
to the negative of the local temperature gradient (heat flows from a hot
reservoir to a cold reservoir):
. We
supplement this with a phenomenological law of heat conduction, known as
Fourier's law:
the heat flux is proportional
to the negative of the local temperature gradient (heat flows from a hot
reservoir to a cold reservoir):
with ![]() the thermal conductivity
of the metal bar. The thermal conductivity is usually measured in units of
the thermal conductivity
of the metal bar. The thermal conductivity is usually measured in units of ![]() , and has dimensions
, and has dimensions ![]() . See the Table on p. 118 of the PQRG for the thermal
conductivities of some materials. Combining Eqs. (1.22) and (1.23), we obtain the
diffusion equation
(often called the heat equation)
. See the Table on p. 118 of the PQRG for the thermal
conductivities of some materials. Combining Eqs. (1.22) and (1.23), we obtain the
diffusion equation
(often called the heat equation)
where ![]() is the thermal diffusivity of the metal bar; it
has dimensions
is the thermal diffusivity of the metal bar; it
has dimensions ![]() , as it should. Eq. (1.24) is the diffusion equation for heat. The diffusion equation will
appear in many other contexts during this course. It usually results from
combining a continuity equation with an empirical law which expresses a
current or flux in terms of some local gradient.
, as it should. Eq. (1.24) is the diffusion equation for heat. The diffusion equation will
appear in many other contexts during this course. It usually results from
combining a continuity equation with an empirical law which expresses a
current or flux in terms of some local gradient.
Suppose that the bar is very long, so that we can consider the idealized
case of an infinite bar. At an initial time t=0, we add an amount of heat H (with dimensions ![]() ) at some point of the bar, which we
will arbitrarily call x=0. We could do this, for instance, by briefly
holding a match to the bar. The heat is conserved at all times, so that
) at some point of the bar, which we
will arbitrarily call x=0. We could do this, for instance, by briefly
holding a match to the bar. The heat is conserved at all times, so that
How does this heat diffuse away from x=0 as a function of time t; i.e.,
what is ![]() ? We first identify the important parameters. The
temperature
? We first identify the important parameters. The
temperature ![]() certainly depends upon x, t, and the diffusivity D; we see from Eq. (1.25) that it also depends upon the initial
conditions through the combination
certainly depends upon x, t, and the diffusivity D; we see from Eq. (1.25) that it also depends upon the initial
conditions through the combination ![]() . What are the
dimensions? We have [x]=L, [t]=T,
. What are the
dimensions? We have [x]=L, [t]=T, ![]() , and
, and ![]() ,
so that n=4. These dimensions are not independent, for the quantity
,
so that n=4. These dimensions are not independent, for the quantity ![]() is dimensionless, so that k=3. We will choose as our
independent quantities (t,D,Q). Now express
is dimensionless, so that k=3. We will choose as our
independent quantities (t,D,Q). Now express ![]() in terms of these
variables:
in terms of these
variables:
We find
which has the solution a=-1/2, b=-1/2, c=1. Therefore, dimensional analysis tells us that the solution of the diffusion equation is of the form
with ![]() a function which we still need to determine. The important
point is that
a function which we still need to determine. The important
point is that ![]() is only a function of the combination
is only a function of the combination ![]() ,
and not x and t separately. To determine
,
and not x and t separately. To determine ![]() , let's introduce the
dimensionless variable
, let's introduce the
dimensionless variable ![]() . Now use the chain rule to calculate
various derivatives of
. Now use the chain rule to calculate
various derivatives of ![]() :
:
Substituting Eqs. (1.30) and (1.31) into the diffusion
equation (1.24), and canceling various factors, we obtain a
differential equation for ![]() ,
,
Dimensional analysis has reduced the problem from the solution of a partial differential equation in two variables to the solution of an ordinary differential equation in one variable! The normalization condition, Eq. (1.25), becomes in these variables
You might think that Eq. (1.32) is hard to solve; however, it turns out that it is an exact differential,
which we can integrate once to obtain
However, since any physically reasonable solution would have both ![]() and
and ![]() as
as ![]() , the
integration constant must be zero. We now need to solve a first order
differential equation, which we do by dividing Eq. (1.35) by
, the
integration constant must be zero. We now need to solve a first order
differential equation, which we do by dividing Eq. (1.35) by ![]() , multiplying by dz, and integrating, with the result that
, multiplying by dz, and integrating, with the result that ![]() , or
, or
with C a constant. To determine C, we use the normalization condition, Eq, (1.33):
where the integral (known as a Gaussian integral) can be found in
integral tables. Therefore ![]() . Returning to our original variables, we have
. Returning to our original variables, we have
This is the complete solution for the temperature distribution in a
one-dimensional bar due to a point source of heat
![]() 2. Here are pictures:
2. Here are pictures:
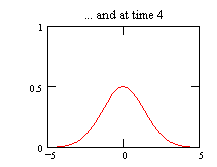
Click here to see an animation (controlled
by mouse buttons) of the one-dimensional diffusion process described
by eq. (1.38). The animation starts at some arbitrary
time t0 (zero time corresponds to an infinitely sharp
peak) and ends at 25t0, and x is in units of the
square root of Dt0.
Footnotes:
![]() 1
I prefer the term diffusion equation, since we are just describing the diffusion of heat.
1
I prefer the term diffusion equation, since we are just describing the diffusion of heat.
![]() 2
For the mathematically sophisticated, I'll mention that the same solution can be obtained using the method of Fourier transforms applied to the diffusion equation.
2
For the mathematically sophisticated, I'll mention that the same solution can be obtained using the method of Fourier transforms applied to the diffusion equation.