The Hydrogen Atom
Michael Fowler, UVa
Factoring Out the Center of Mass Motion
The hydrogen atom consists of two particles, the proton and the electron, interacting via the Coulomb potential , where as usual . Writing the masses of the two particles as Schrödinger’s equation for the atom is:
But are not the most natural position variables for describing this system: since the potential depends only on the relative position, a better choice is defined by:
so is the center of mass of the system. It is convenient at the same time to denote the total mass by and the reduced mass by
Transforming in straightforward fashion to the variables Schrödinger’s equation becomes
Writing the wave function
we can split the equation into two:
and the total system energy is Note that the motion of the center of mass is (of course) just that of a free particle, having a trivial plane wave solution. From now on, we shall only be concerned with the relative motion of the particles. Since the proton is far heavier than the electron, we will almost always ignore the difference between the electron mass and the reduced mass, but it should be noted that the difference is easily detectable spectroscopically: for example, the lines shift if the proton is replaced by a deuteron (heavy hydrogen).
We’re ready to write Schrödinger’s equation for the hydrogen atom, dropping the suffixes in the second equation above, and writing out explicitly in spherical coordinates:
Factoring Out the Angular Dependence: the Radial Equation
Since the potential is spherically symmetric, the Hamiltonian commutes with the angular momentum operators so we can construct a common set of eigenkets of the three operators The angular dependence of these eigenkets is therefore that of the ’s, so the solutions must be of the form
Now, notice that in the Schrödinger equation above, the angular part of is exactly the differential operator , so operating on it will give .
The spherical harmonic can then be cancelled from the two sides of the equation leaving:
it now being apparent that cannot depend on
The radial derivatives simplify if one factors out from the function writing
and temporarily suppressing the and to reduce clutter.
The equation becomes:
Rearranging,
Note that this is the same as the Schrödinger equation for a particle in one dimension, restricted to , in a potential (for ) going to positive infinity at the origin, then negative and going to zero at large distances, so it always has a minimum for some positive
We are interested in bound states of the proton-electron system, so will be a negative quantity. At large separations, the wave equation simplifies to
having approximate solutions
The bound states we are looking for, of course, have exponentially decreasing wave functions at large distances.
Going to a Dimensionless Variable
To further simplify the equation, we introduce the dimensionless variable
giving
where (for reasons which will become apparent shortly) we have introduced defined by
Notice that in transforming from to the dimensionless variable the scaling factor depends on energy, so will be different for different energy bound states!
Consider now the behavior of the wave function near the origin. The dominant term for sufficiently small is the centrifugal one, so
for which the solutions are Since the wave function cannot be singular, we choose the second.
We have established that the wave function decays as at large distances, and goes as close to the origin. Factoring out these two asymptotic behaviors, define by
It is straightforward (if tedious!) to establish that satisfies the differential equation:
Putting in a trial series solution gives a recurrence relation between successive coefficients:
For large values of , so and therefore . This means we have found the diverging radial wavefunction , which is in fact the correct behavior for general values of the energy.
To find the bound states, we must choose energies such that the series is not an infinite one. As long as the series stops somewhere, the exponential decrease will eventually take over, and yield a finite (bound state) wave function. Just as for the simple harmonic oscillator, this can only happen if for some Inspecting the ratio , evidently the condition for a bound state is that
in which case the series for terminates at From now on, since we know that for the functions we’re interested in is an integer, we replace by
To find the energies of these bound states, recall and , so
so
(This defines the Rydberg, a popular unit of energy in atomic physics.)
Remarkably, this is the very same series of bound state energies found by Bohr from his model! Of course, this had better be the case, since the series of energies Bohr found correctly accounted for the spectral lines emitted by hot hydrogen atoms. Notice, though, that there are some important differences with the Bohr model: the energy here is determined entirely by called the principal quantum number, but, in contrast to Bohr’s model, is not the angular momentum. The true ground state of the hydrogen atom, has zero angular momentum: since , means both and The ground state wave function is therefore spherically symmetric, and the function is just a constant. Hence and the actual radial wave function is this divided by and of course suitably normalized.
To write the wave function in terms of we need to find (the energy-dependent scaling factor we used in going to a dimensionless variable). Putting together , and ,
where the length
is called the Bohr radius: it is in fact the radius of the lowest orbit in Bohr’s model.
Exercise: check this last statement.
It is worth noting at this point that the energy levels can be written in terms of the Bohr radius a0:
(This is actually obvious: remember that the energies are identical to those in the Bohr model, in which the radius of the orbit is so the electrostatic potential energy is etc.)
Moving on to the excited states: for we have a choice: either the radial function can have one term, as before, but now the angular momentum (since ); or can have two terms (so ), and Both options give the same energy, -0.25 Ry, since is the same, and the energy only depends on
In fact, there are four states at this energy, since has states with and and has the one state (For the moment, we are not counting the extra factor of 2 from the two possible spin orientations of the electron.)
For there are 9 states altogether: gives one, gives 3 and gives 5 different values. In fact, for principal quantum number there are degenerate states. ( being the sum of the first odd integers.)
The states can be mapped out, energy vertically, angular momentum horizontally:
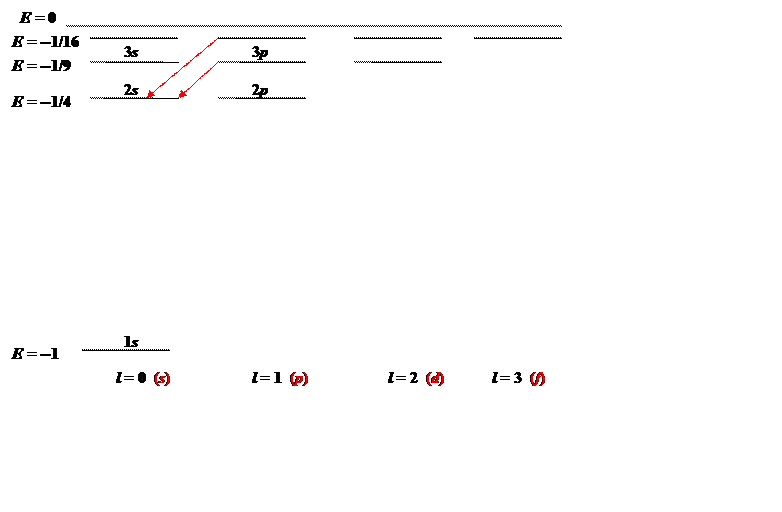
The energy the levels are labeled being the principal quantum number and the traditional notation for angular momentum is given at the bottom of the diagram. The two red vertical arrows are the first two transitions in the spectroscopic Balmer series, four lines of which gave Bohr the clue that led to his model. The corresponding series of transitions to the ground state are in the ultraviolet, they are called the Lyman series.
Wave Functions for some Low-n States
From now on, we label the wave functions with the quantum numbers, , so the ground state is the spherically symmetric .
For this state , where , with a constant, and .
So, as a function of with an easily evaluated normalization constant:
For the function is still a single term, a constant, but now , and, for , remembering the energy-dependence of
Therefore . Again, evaluating the normalization constant is routine, yielding
.
The wave functions for the other -values, have the in replaced by respectively (from the earlier discussion of the ’s).
The other state has so from we have and the series for has two terms, and the ratio being
for the relevant values: So . For , the normalized wave function is
Note that the zero angular momentum wave functions are nonzero and have nonzero slope at the origin. This means that the full three dimensional wave functions have a slope discontinuity there! But this is finethe potential is infinite at the origin. (Actually, the proton is not a point charge, so really the kink will be smoothed out over a volume of the size of the protona very tiny effect.)
General Solution of the Radial Equation
In practice, the first few radial functions can be constructed fairly easily using the method presented above, but it should be noted that the differential equation for
is in fact Laplace’s equation, usually written
where are integers, and is a Laguerre polynomial (Messiah, page 482).
The two equations are the same if z = 2ρ, and the solution to the radial equation is therefore
Quoting Messiah, the Laguerre polynomials , and the associated Laguerre polynomials are given by:
(These representations can be found neatly by solving
Laplace’s equation using surprise a
But what do they look like? The function is zero at the origin (apart from the trivial case ) and zero at infinity, always positive and having nonzero slope except at its maximum value, . The derivatives bring in separated zeroes, easily checked by sketching the curves generated by successive differentiation. Therefore, , a polynomial of degree has real positive zeroes, and value at the origin , since the only nonzero term at is that generated by all differential operators acting on .
The associated Laguerre polynomial is generated by differentiating times. Now has real positive zeroes, differentiating it gives a polynomial one degree lower, with zeroes which must be one in each interval between the zeroes of . This argument remains valid for successive differentiations, so must have p real separate zeroes.
Putting all this together, and translating back from to the radial solutions are:
with the normalization constant.
Here are the three radial wave functions:

The number of nodes, the radial quantum number, is (Note: The relative normalizations are correct here, but not the overall normalization.)
For higher values, the wave functions become reminiscent of classical mechanics. for example, for the highest angular momentum state probability distribution peaks at , the Bohr orbit radius:
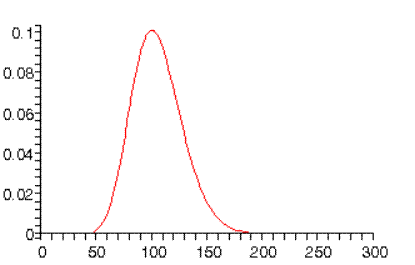
whereas for we find:
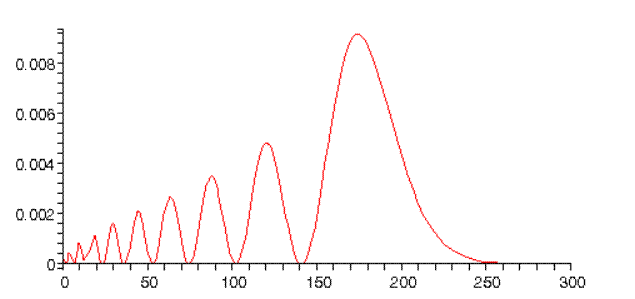
Notice this peaks just below twice the Bohr radius. This can be understood from classical mechanics: for an inverse square force law, elliptical orbits with the same semimajor axis have the same energy. The orbit is a circle, the orbit is a long thin ellipse (one end close to the proton), so it extends almost twice as far from the origin as the circle. Furthermore, the orbiting electron will spend longer at the far distance, since it will be moving very slowly.
(Note: the normalizations in the above graphs are only approximate.)